Contents
|
What is Nitrosative Stress?
Whilst terms such as “antioxidants”, “free radicals” and even “oxidative stress” are now widely used; most people have never heard of “nitrosative stress”, despite its major heath significance.
Nitrosative (or nitro) stress is a form of oxidative stress – yet here it is not oxygen radicals that damage the body, but an excessive production of nitrogen monoxide radicals (chemical abbreviation: NO). Through an oversupply of NO radicals and the increased formation of dangerous secondary products – such as the highly toxic peroxynitrite – cells, mebranes and DNA can be damaged and a whole range of illnesses and symptoms can occur (1, 2).
Vitamin B12, particularly in the form of hydroxocobalamin, is the natural opponent of NO and renders it harmless in the body. One of the many benefits of vitamin B12 is that it is a very effective NO scavenger and is successfully administers to fight nitro stress (3, 4).
Key Points
|
NO Radicals: Friend or Foe?
As is so often the case with the careful chemical balances within the body, in the case of nitrosative stress it is the dose that makes the poison: nitrogen monoxide is actually an important endogenous substance that fulfils numerous biological functions. NO relaxes the blood vessels, lowers blood pressure, is part of the body’s immune system and even acts as a messenger substance in the nervous system.
However, the NO level must be tightly regulated by the body through various systems and held within a very narrow window. Nitrosative stress occurs when there is an excessive amount of NO radicals in the body. These radicals and their derivatives are highly dangerous as they knocks multiple biological systems out of balance. As a result, researchers have identified nitro stress as the cause of numerous clinical pictures (5-7).
The Consequences of Nitrosative Stress
NO radicals are highly active biologically, they can penetrate all biological membranes and take effect on the entire physical systems very quickly. Unlike other messenger substances, NO radicals do not dock to receptors, but bind directly to various enzymes inside the cells, thereby inhibiting their function. Plus, they alter the membrane potentials of cells and cell organelles, opening certain membrane channels. This causes a drastic change of the chemistry within the cell, with various consequences for the cell metabolism.
Through the oversupply of NO radicals, the highly reactive peroxynitrite is increasingly created, which inhibits further enzymes (8) and causes severe damage to cells (9, 10). Nitrosative stress can consequently trigger a whole range of negative symptoms.
NO Stress
- Inhibits energy production in the mitochondria (11 – 15)
- Disrupts important detoxification enzymes
- Consumes important antioxidants (in particular, melatonin and glutathione) (16 – 18)
- Disturbs hormone synthesis (19)
- Disrupts neurotransmitter synthesis (20)
- Leads to DNA damage (21 -24)
- Interferes with the cell cycle and leads to the death of cells (25, 26)
- Disturbs the haem system (iron-containing enzyme) (27 – 29)
- Disrupts the breakdown of cholesterol
Researchers of alternative medicine such as Professor Martin Pall, Dr Heinrich Kremer and Dr Bodo Kuklinski are all of the opinion that nitrosative stress is one of the most crucial variables in the emergence of chronic diseases. It is thus a matter of urgency that anyone who suspects themselves to be experiencing nitrosative stress conducts the appropriate tests and starts taking countermeasures (see below).
Nitrosative Stress, Tryptophan and Tyrosine
Through nitro stress and its derived product peroxynitrite, the important amino acids of tryptophan and tyrosine are rendered ineffectual. These two amino acids however play a crucial role in the synthesis of further neurotransmitters and hormones:
| Amino Acid | Neurotransmitters |
| Tryptophan | Serotonin, melatonin |
| Tyrosine | Dopamine, adrenalin, noradrenalin, melanin |
When production of these neurotransmitters is blocked, it leads to a whole range of symptoms: serotonin and dopamine are considered to be “happy hormones” because of their major impact on mood, motivation and psychological stability. Serotonin and melatonin regulate sleeping patterns. Together these neurotransmitters have an enormous influence on blood pressure and the central nervous system.
NO Stress and Damage to the Mitochondria
The energy powerhouses of the cells, the mitochondria, are particularly vulnerable to nitrosative stress. Mitochondria are controlled by a particular genome, which is 10 times more sensitive to NO stress than all cell DNA. While many of the consequences of nitrosative stress are reversible, if exposure is long term, irreversible damage to certain enzyme functions and in the genome of the mitochondria can occur, which can even be inherited maternally (30).
Such mitochondriopathies – either through maternal inheritance or as the consequence of a self-acquired mutation of the mitochondrial DNA – are becoming increasingly common (31, 32). Nitrosative and oxidative stress are probably the main causes of these diseases.
NO Stress: The Cause of Many Chronic Diseases?
For the first time, the biochemical processes resulting from NO stress can be used to conclusively explain various diseases in chemical terms, some of which have remained a mystery until recently. These include:
- Chronic fatigue syndrome or myalgic encephalomyelitis (CFS/ME) (33, 34)
- Fibromylagia (35 – 37)
- Multiple sclerosis (38, 39)
- Rheumatoid arthritis (40)
- Multiple chemical sensitivity or idiopathic environmental intolerances (MCS/IEI) (41)
- Post-traumatic stress disorder (PTSD)
- Irritable bowel syndrome (IBS) (42)
- Certain forms of Parkinson’s disease (43)
- Psoriasis, neurodermatitis (44)
- Atherosclerosis (45, 46)
- Allergies (47, 48)
- Chronic recurrent infections
- Chronic anxiety and depression
- Chronic insomnia
Multiple therapists worldwide have reported very good treatment results based of the connection between NO stress and the above-mentioned diseases. Plus, clinical studies which confirm this postulate are on the increase. Nevertheless, the theory of nitro stress as the cause of these diseases remains the subject of scientific debate and has not yet gained general recognition.
Vitamin B12 and NO Stress
The theory of Professor Pall and his colleagues not only explains conclusively for the first time how these diseases develop, but also why vitamin B12 has a good effect in almost all cases. B12 is the natural opponent of NO radicals and simultaneously an important part of the body’s own regulation system for nitric oxide. Through a variety of mechanisms, the vitamin ensures both the controlled formation, as well as the inhibition and deactivation of NO (49, 50). B12 is therefore a central regulator of the body, keeping the NO level within the biologically desired range (51, 52).
Nitrosative Stress and Hydroxocobalamin
Besides its function in the regulation of NO formation, vitamin B12 is additionally an effective NO scavenger. The B12 form of hydroxocobalamin in particular binds reliably to excessive NO radicals and neutralises them. Cyanocobalamin, methylcobalamin and adenosylcobalamin, on the other hand, show far less activity with respect to the neutralisation of NO radicals (53, 54). This action is most probably a central function of hydroxocobalamin, which accounts for around 50% of the free vitamin B12 in the blood (55).
In addition to hydroxocobalamin, reduced cobalamin – a biological intermediate form – is particularly reactive to NO radicals (56). It cannot yet be used directly for medical purposes, however, and thus hydroxocobalamin is currently regarded as the best vitamin B12 form for nitro stress.
Nitrosative Stress and Vitamin B12 Deficiency
Vitamin B12 administered for the detoxification of NO radicals is oxidised and largely excreted; therefore no longer available for its further vitamin functions. Nitrosative stress can therefore be the catalyst of a vitamin B12 deficiency, since a high exposure to NO consumes large amounts of the vitamin. Accordingly, the vitamin B12 dosages required to treat nitro stress are higher than the daily requirement; injections or very high oral doses are often used.
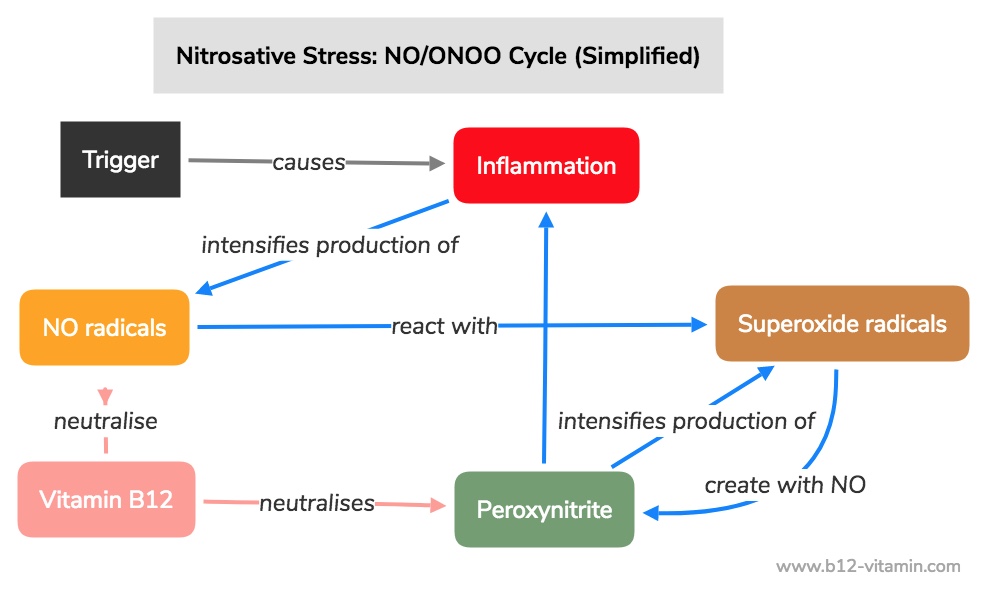
Nitrosative Stress and Peroxynitrite: the NO/ONOO– Cycle
A large component of the dangerous effects of nitrosative stress are not due to the NO radicals themselves, but due to the secondary product, peroxynitrite (57, 58). Peroxynitrite is created through a reaction between NO and the oxygen radical, superoxide:
Nitrogen monoxide + superoxide = peroxynitrite NO + O2- = ONOO-
This reaction was dubbed the NO/ONOO- cycle by Professor Martin Pall because it is a self-reinforcing, chronic vicious circle: the excessive supply of NO radicals leads to an increased formation of the highly toxic peroxynitrite; this peroxynitrite in turn promotes the formation of NO radicals and superoxide, which combine to form even more peroxynitrite.
Through inflammations, the production of NO and superoxide increases by a thousand times, which subsequently leads to a million-fold increase in the formation of peroxynitrite (59).
What Causes the Nitro Stress Vicious Circle?
This vicious circle is triggered by various factors that cause the body’s own NO production to increase:
- Viral and bacterial infections/chronic inflammations
- Exposure to toxins (heavy metals, pesticides, chemicals)
- Medications (antibiotics, cytostatic drugs, statins, long-term nitrates, sexual enhancers)
- Physical traumas (especially whiplash or relating to the spine)
- Extreme psychological stress
- Extreme physical strain
- Oxygen deprivation
Once triggered, the chronic cycle persists even if the causal trigger has long since disappeared. It cannot be eliminated with conventional therapies, as these usually only target individual symptoms with medicines, which often even aggravate the cycle.
Nitrate and NO Radicals
The NO/ONOO- vicious circle is further inflamed by the fact that many conventional foods are contaminated with large quantities of nitrite and nitrate: fertiliser, pesticides, conservatives, plasticisers, artificial colourings and multiple other chemical additives containing unnaturally high amounts of nitrogen compounds. We also absorb nitrogen compounds through the skin via cosmetics, colourants and textile additives; and even through the air, where pollution is almost ubiquitous.
Diagnosing Nitrosative Stress
Nitro stress can be determined through a simple urine test, in which certain parameters are measured:
| Substance | Mechanism | Normal Value |
| Citrulline | Bi-product in the creation of NO radicals | < 2.9 mg/g creatinine |
| Nitrophenylacetic acid | Secondary product of NO stress | < 3.0 µg/g creatinine |
| Methylmalonic acid | Signals a B12 deficiency in the mitochondria, indication of NO stress | < 2 mg/g creatinine |
Urine tests can be conducted by both doctors and naturopaths. Home testing kits are also available online.
Therapy: Treating Nitrosative Stress
Nitrosative stress is rarely diagnosed by GPs and many patients are simply sent home with the assertion that their symptoms are “psychosomatic”. This often leads to years of chronic suffering or even false diagnoses and treatments. This is all the more unfortunate because today nitrosative stress can be treated quite easily with nutrients alone. Once the trigger has been removed, it is necessary to break the vicious circle and replenish the nutrients consumed during its course. The therapy has three important elements:
- Treatment of the trigger
- Avoidance of all factors that could further fuel the cycle
– Conventionally grown vegetables
– Smoked foods
– Cigarette smoke
– Glutamate and aspartate
– Medicines, if possible
– Stress and strain (psychological and physical)
– Conventional cosmetics - Breaking the cycle
– High dose vitamin B12
– Vitamin mix: B vitamins, vitamin C, E, A and D
– Minerals and trace elements: zinc, selenium, magnesium, manganese, copper
– Nutrients: alpha lipoic acid, bioflavonoids, cysteine, turmeric, omega-3 fatty acids, Q10
– Hormones/antioxidants: melatonin, glutathione
A combination of vitamin B12 (hydroxocobalamin), turmeric and green tea is ideal for home use. In severe cases, however, intravenous (IV) therapy is sometimes necessary in order to effectively break the cycle.
Conclusion: Nitrosative Stress
The new findings on nitrosative stress could trigger a paradigm shift in the treatment of many diseases, which previously could not be explained in medical terms and were either falsely treated or not treated at all – often alleged to be “psychosomatic” illnesses. The decoding of these biochemical mechanisms provides for the first time a solid theoretical basis for the explanation and treatment of these diseases and also for the versatile, therapeutic and stabilising effects of vitamin B12. As a natural antagonist of NO radicals, B12 is a central therapeutic agent in this context, likely to become even more important over the coming years.
Sources
- Beckman JS, Koppenol WH. Nitric oxide, superoxide, and peroxynitrite: the good, the bad, and ugly. Am J Physiol Cell Physiol 271: C1424–C1437, 1996.
- Squadrito GL, Pryor WA. Oxidative chemistry of nitric oxide: the roles of superoxide, peroxynitrite, and carbon dioxide. Free Radic Biol Med 1998; 25:392–403.
- Martin L. Pall. Cobalamin Used in Chronic Fatigue Syndrome Therapy Is a Nitric Oxide Scavenger. Journal of Chronic Fatigue Syndrome Vol. 8, Iss. 2, 2000
- van der Kuy PH, Merkus FW, Lohman JJ, ter Berg JW, Hooymans PM. Hydroxocobalamin, a nitric oxide scavenger, in the prophylaxis of migraine: an open, pilot study. Cephalalgia. 2002 Sep;22(7):513-9
- Pall, M. L.: Explaining „Unexplained Illnesses“: Disease Paradigm for Chronic Fatigue Syndrome, Multiple Chemical Sensitivity, Fibromyalgia, Post-Traumatic Stress Disorder, Gulf War Syndrome, and others. HPP (2007): ISBN 978-0-7890-2389-6.
- Kuklinski, B.: Nitrosativer Stress, Mitochondriopathie und Mikronährstoffe. Ärztekammer Baden-Württemberg (2006)
- Kuklinski, B.: Praxisrelevanz des nitrosativen Stresses. Umwelt Med. Gesellsch. 18. (2005) S. 95.
- Alvarez B, Radi R. Peroxynitrite reactivity with amino acids and proteins. Amino Acids. 2003 Dec;25(3-4):295-311. Epub 2003 Sep 26. Review.
- Beckman JS. Oxidative damage and tyrosine nitration from peroxynitrite. Chem Res Toxicol 9: 836–844, 1996.
- Estevez AG, Spear N, Pelluffo H, Kamaid A, Barbeito L, Beckman JS. Examining apoptosis in cultured cells after exposure to nitric oxide and peroxynitrite. In: Nitric Oxide: Biological and Antioxidant Activities, edited by Packer L. San Diego, CA: Academic, 1998, p. 393–402.
- Radi R, Cassina A, Hodara R. Nitric oxide and peroxynitrite interactions with mitochondria. Biol Chem 383: 401–409, 2002.
- Radi R, Cassina A, Hodara R. Nitric oxide and peroxynitrite interactions with mitochondria. Biol Chem 383: 401–409, 2002.
- Brookes PS, Bolanos JP, Heales SJ. The assumption that nitric oxide inhibits mitochondrial ATP synthesis is correct. FEBS Lett 446: 261–263, 1999.
- Radi R, Rodriguez M, Castro L, Telleri R. Inhibition of mitochondrial electron transport by peroxynitrite. Arch Biochem Biophys 308: 89–95, 1994.
- Cleeter MWJ, Cooper JM, Darley-Usmar VM, Moncada S, Schapira AHV. Reversible inhibition of cytochrome c oxidase, the terminal enzyme of the mitochondrial respiratory chain, by nitric oxide. FEBS Lett 345: 50–54, 1994.
- Arteel GE, Briviba K, Sies H. Protection against peroxynitrite. FEBS Lett 445: 226–230, 1999.
- Galano, A., Tan, D. X. and Reiter, R. J. (2011), Melatonin as a natural ally against oxidative stress: a physicochemical examination. Journal of Pineal Research, 51: 1–16.
- Noda Y, Mori A, Liburdy R et al. Melatonin and its precursors scavenge nitric oxide. J Pineal Res 1999; 27:159–163.
- Blanchard-Fillion B, Souza JM, Friel T, Jiang GC, Vrana K, Sharov V, Barron L, Schoneich C, Quijano C, Alvarez B, Radi R, Przedborski S, Fernando GS, Horwitz J, Ischiropoulos H. Nitration and inactivation of tyrosine hydroxylase by peroxynitrite. J Biol Chem 276: 46017–46023, 2001.
- Park S, Geddes TJ, Javitch JA, Kuhn DM. Dopamine prevents nitration of tyrosine hydroxylase by peroxynitrite and nitrogen dioxide: is nitrotyrosine formation an early step in dopamine neuronal damage? J Biol Chem 278: 28736–28742, 2003.
- Burney S, Caulfield JL, Niles JC, Wishnok JS, Tannenbaum SR. The chemistry of DNA damage from nitric oxide and peroxynitrite. Mutat Res 424: 37–49, 1999.
- Wink D, Kasprzak K, Maragos C, Elespuru R, Misra M, Dunams T, Cebula T, Koch W, Andrews A, Allen J, Keefer L. DNA deaminating ability and genotoxicity of nitric oxide and its progenitors. Science 254: 1001–1003, 1991.
- Szabó C. Poly(ADP-ribose) polymerase activation by reactive nitrogen species–relevance for the pathogenesis of inflammation. Nitric Oxide. 2006 Mar;14(2):169-79. Epub 2005 Aug 18. Review.
- Wiseman H, Halliwell B. Damage to DNA by reactive oxygen and nitrogen species: role in inflammatory disease and progression to cancer. Biochem J. 1996 Jan 1;313 ( Pt 1):17-29. Review.
- Virag L, Szabo E, Gergely P, Szabo C. Peroxynitrite-induced cytotoxicity: mechanism and opportunities for intervention. Toxicol Lett 140–141: 113–124, 2003.
- Bonfoco E, Krainc D, Ankarcrona M, Nicotera P, Lipton SA. Apoptosis and necrosis: two distinct events induced, respectively, by mild and intense insults with N-methyl-d-aspartate or nitric oxide/superoxide in cortical cell cultures. Proc Natl Acad Sci USA 92: 7162–7166, 1995.
- Boccini F, Herold S. Mechanistic studies of the oxidation of oxyhemoglobin by peroxynitrite. Biochemistry 43: 16393–16404, 2004.
- Herold S, Exner M, Boccini F. The mechanism of the peroxynitrite-mediated oxidation of myoglobin in the absence and presence of carbon dioxide. Chem Res Toxicol 16: 390–402, 2003.
- Thomson L, Trujillo M, Telleri R, Radi R. Kinetics of cytochrome c oxidation by peroxynitrite: implications for superoxide measurements in nitric oxide-producing biological systems. Arch Biochem Biophys 319: 491–497, 1995.
- Sperl, W. 23.11 Mitochondriale Erkrankungen (Mitochondriopathien)
- Sanderson S, Green A, Preece MA, Burton H (2006) The in- cidence of inherited metabolic disorders in the West Midlands. UK. Arch Dis Child 91: 896 – 9
- Schaefer AM, Taylor RW, Turnbull DM, Chinnery PF (2004) The epidemiology of mitochondrial disorders – past, present and future. Biochim Biophys Acta 1 6 5 9 : 1 1 5 – 2 0
- Pall, M. L. Elevated, sustained peroxynitrite levels as the cause of chronic fatigue syndrome. Medical hypotheses, 2000, 54. Jg., Nr. 1, S. 115-125
- Smirnova, Iva V.; PALL, Martin L. Elevated levels of protein carbonyls in sera of chronic fatigue syndrome patients. Molecular and cellular biochemistry, 2003, 248. Jg., Nr. 1-2, S. 93-95.
- Pall, Martin L. The NO/ONOO–cycle as the cause of fibromyalgia and related illnesses: etiology, explanation and effective therapy. New Research in Fibromyalgia, 2006, 2. Jg., S. 39-59.
- Ozgocmen, Salih, et al. Current concepts in the pathophysiology of fibromyalgia: the potential role of oxidative stress and nitric oxide. Rheumatology international, 2006, 26. Jg., Nr. 7, S. 585-597.
- Meeus, Mira, et al. The role of mitochondrial dysfunctions due to oxidative and nitrosative stress in the chronic pain or chronic fatigue syndromes and fibromyalgia patients: peripheral and central mechanisms as therapeutic targets?. Expert opinion on therapeutic targets, 2013, 17. Jg., Nr. 9, S. 1081-1089.
- Bo Liu, Yuefei Shen, Kaiwen Xiao, Yulan Tang, Luan Cen, and Junjie Wei. “Serum uric acid levels in patients with multiple sclerosis: a meta-analysis.” Neurological Research 2012; 34(2), 163-171.
- Hooper DC, Spitsin S, Kean RB, Champion JM, Dickson GM, Chaudhry I, Koprowski H. Uric acid, a natural scavenger of peroxynitrite, in experimental allergic encephalomyelitis and multiple sclerosis. Proc Natl Acad Sci U S A. 1998 Jan 20;95(2):675-80.
- Migita K. Nitric oxide in rheumatoid arthritis. Nippon Rinsho 63 Suppl 1: 106–109, 2005.
- Pall, Martin L., and James D. Satterlee. “Elevated nitric oxide/peroxynitrite mechanism for the common etiology of multiple chemical sensitivity, chronic fatigue syndrome, and posttraumatic stress disorder.” Annals of the New York Academy of Sciences 933.1 (2001): 323-329.
- Dijkstra G, Moshage H, van Dullemen HM, de Jager-Krikken A, Tiebosch AT, Kleibeuker JH, Jansen PL, van Goor H. Expression of nitric oxide synthases and formation of nitrotyrosine and reactive oxygen species in inflammatory bowel disease. J Pathol 186: 416–421, 1998.
- Zhang L, Dawson VL, Dawson TM. Role of nitric oxide in Parkinson’s disease. Pharmacol Ther 109: 33–41, 2006.
- Sikar Aktürk, A., Özdogan, H.K., Bayramgürler, D., Çekmen, M.B., Bilen, N. and Kiran, R. (2012), Nitric oxide and malondialdehyde levels in plasma and tissue of psoriasis patients. Journal of the European Academy of Dermatology and Venereology, 26: 833–837.
- Hogg N, Darley-Usmar VM, Graham A, Moncada S. Peroxynitrite and atherosclerosis. Biochem Soc Trans 21: 358–362, 1993.
- Huige Li, Sven Horke, Ulrich Förstermann, Vascular oxidative stress, nitric oxide and atherosclerosis, Atherosclerosis, Volume 237, Issue 1, November 2014, Pages 208-219, ISSN 0021-9150
- Russell P. Bowler, James D. Crapo, Oxidative stress in allergic respiratory diseases, Journal of Allergy and Clinical Immunology, Volume 110, Issue 3, September 2002, Pages 349-356, ISSN 0091-6749
- Gabr, Sami A.; Al-Ghadir, Ahmad H. Role of cellular oxidative stress and cytochrome c in the pathogenesis of psoriasis. Archives of dermatological research, 2012, 304. Jg., Nr. 6, S. 451-457.
- Carmen Wheatley. The return of the Scarlet Pimpernel: cobalamin in inflammation II — cobalamins can both selectively promote all three nitric oxide synthases (NOS), particularly iNOS and eNOS, and, as needed, selectively inhibit iNOS and nNOS. Journal of Nutritional & Environmental Medicine Volume 16, Issue 3-4, 2007
- Vijay S. Sharma et al. Reactions of Nitric Oxide with Vitamin B12 and Its Precursor, Cobinamide. Biochemistry 2003 42 (29), 8900-8908
- Carmen Wheatley, (2007) Cobalamin in inflammation III — glutathionylcobalamin and methylcobalamin/adenosylcobalamin coenzymes: the sword in the stone? How cobalamin may directly regulate the nitric oxide synthases. Journal of Nutritional & Environmental Medicine 16:3-4, pages 212-226.
- André L. F. Sampaio, Jesmond Dalli, Vincenzo Brancaleone, Fulvio D’Acquisto, Mauro Perretti, and Carmen Wheatley, “Biphasic Modulation of NOS Expression, Protein and Nitrite Products by Hydroxocobalamin Underlies Its Protective Effect in Endotoxemic Shock: Downstream Regulation of COX-2, IL-1, TNF-, IL-6, and HMGB1 Expression,” Mediators of Inflammation, vol. 2013, Article ID 741804, 20 pages, 2013.
- J. Brice Weinberg, Youwei Chen, Ning Jiang, Bethany E. Beasley, John C. Salerno, Dipak K. Ghosh, Inhibition of nitric oxide synthase by cobalamins and cobinamides, Free Radical Biology and Medicine, Volume 46, Issue 12, 15 June 2009, Pages 1626-1632, ISSN 0891-5849
- M Brouwer, W Chamulitrat, G Ferruzzi, DL Sauls, JB Weinberg. Nitric oxide interactions with cobalamins: biochemical and functional consequences. Blood Sep 1996, 88 (5) 1857-1864
- J. van Kapel, L.J.M. Spijkers, J. Lindemans, J. Abels, Improved distribution analysis of cobalamins and cobalamin analogues in human plasma in which the use of thiol-blocking agents is a prerequisite, Clinica Chimica Acta, Volume 131, Issue 3, 15 July 1983, Pages 211-224, ISSN 0009-8981
- Kruszyna, Harriet, et al. Spectroscopic studies of nitric oxide (NO) interactions with cobalamins: reaction of NO with superoxocobalamin (III) likely accounts for cobalamin reversal of the biological effects of NO. Journal of Pharmacology and Experimental Therapeutics, 1998, 285. Jg., Nr. 2, S. 665-671.
- Huie RE, Padmaja S. The reaction rate of nitric oxide with superoxide. Free Rad Res Commun 18: 195–199, 1993
- Squadrito GL, Pryor WA. The formation of peroxynitrite in vivo from nitric oxide and superoxide. Chem Biol Interact 1995; 96:203–206.
- Pál Pacher, Joseph S. Beckman, Lucas Liaudet. Nitric Oxide and Peroxynitrite in Health and Disease. Physiological Reviews Published 1 January 2007 Vol. 87 no. 1, 315-424